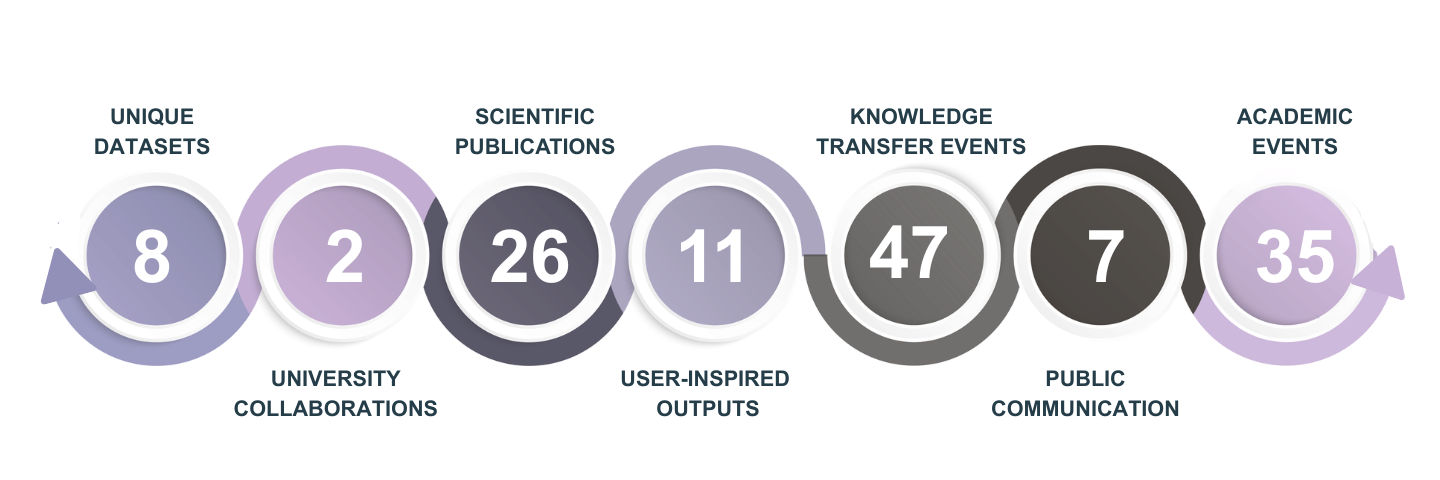
Research work conducted and main research results
The original objective of the project was to deepen understanding of the political, economic and organizational factors required to implement alternative innovation models (AIMs) of pharmaceutical R&D that could jointly achieve innovation and global access to medicines. By its conclusion, the project had clarified the concept of alternative pharmaceutical innovation models, substantially expanded the evidence base on such models, increased understanding of the diversity and functioning of such models, and developed a conceptual framework of how such models co-exist with traditional innovation models in niches within the broader innovation ecosystem, and a theory of what would be required to scale-up further and/or to institutionalize them. Throughout the project, we iteratively engaged with practitioners and policymakers.